Minerals used in everyday life are more common than you think. Many are unaware of what goes into the “built environment” surrounding them, how firmly it is linked to the mineral world, and what those natural products looked like before being melted, smelted and “svelted” into manufactured goods.

Minerals used in everyday life are all around us and even within us. Consider iron in your blood or calcium in your bones. While humans have always sought food products, we’ve always used rocks. Our ancestors used cobbles as hammers to crack nuts. We later learned to use obsidian, chert, and flint to knap knives and spear points and to spark fire. We’ve moved on from the Stone Age to the Copper, Bronze, and Iron Ages. Minerals and related earth resources continue to enable contemporary life and the built environment in which we live. They include metals, nonmetallic minerals, and fossil fuels. How little we appreciate this fact of life!
Minerals Used in Everyday Life: The Pencil Example
Something as simple as a pencil requires more minerals than you might imagine. While a pencil casing is painted wood with a hollow core, the rod within the core is a combination of graphite (carbon) and kaolinite (clay). The more kaolinite, the harder the rod. This is why we have #2, #3, and other pencil grades that leave either a wide dark streak or a slender light streak. While the pencil eraser is a natural or synthetic rubber, it may contain pumice to provide grit. Holding that eraser to the pencil is a tube constructed of aluminum (from bauxite) or brass (from copper plus zinc, or sphalerite). Four to six minerals in a common pencil. Who knew?!
To appreciate the number of minerals used in everyday life, deconstruct other objects. A salt shaker often has an aluminum top (derived from bauxite) and a glass body (from sand, or silicon dioxide) and is filled with salt (halite) crystals. Although it’s on its way out, an old-fashioned incandescent light bulb has a glass exterior (made from silica, soda ash, lime, coal, and salt), a brass or aluminum screw-in base, a tungsten filament, copper and nickel lead-in wires, molybdenum tie and support wires, and an aluminum heat deflector.
Minerals Used in Everyday Life That You Can Collect EasilyIn building a collection of earth resources, the following can easily be found at a gem show or rock shop: calcite, copper, feldspar, fluorite, galena, garnet, halite, hematite, quartz, and sulfur. Some serve double duty. For instance, calcite, feldspar, fluorite, and quartz can be used to form a collection of Mohs’ Scale minerals. |
Minerals That Shape Our Built Environment
Here are some fun and easy exercises to introduce kids (and yourself ) to the many minerals contributing to our built environment.

Match the Product to the Mineral
An Interactive Display & Quiz
Perfect for a school project…Construct an interactive display showing everyday items at the back and the minerals that went into them at the front. For instance, a soda can at the back and a specimen of bauxite (aluminum ore) at the front, or matches at the back and sulfur at the front. Provide a quiz for kids to fill out to match a mineral to a product.
Spin the Wheel!
For more immediate interactive fun, have a board laid out with squares numbered and stocked with different economic minerals. Kids spin the wheel. They then need to name a product made from a mineral on the number where the wheel lands. If they guess correctly, they keep the mineral. Stick with fairly easy and obvious choices (e.g., a copper nugget matched to plumbing pipes) and have a poster or chart nearby that kids can consult.
Minerals Used in Everyday Life: Inside Your SmartphoneIf you were surprised to learn four to six minerals are contained in a pencil, that’s nothing! That smartphone in your pocket? It may contain copper, silver (from argentinite), gold, palladium, platinum, arsenic (from realgar), gallium, magnesium, tungsten (from scheelite), petroleum products, nickel, quartz (silica), halite, cassiterite (tin ore), bauxite (aluminum ore), chromite, sphalerite, chalcopyrite, tetrahedrite, arsenopyrite, tantalite, wolframite, spodumene or lepidolite (lithium ores), graphite, bastnaesite, sylvite, columbite, titanium (from rutile), monzanite (an ore of neodymium) and more! |
Discover Minerals in Everyday Household Items

In a school classroom, rock club meeting room, or a home, gather kids around a flipchart, chalkboard, or whiteboard. Encourage them to look around and list everyday things and the rocks and minerals that went into them. If using an old-fashioned chalkboard, you can start with the chalk and the slate of the chalkboard. You might go throughout an entire house, or focus on a particular room.
Here are just a few examples: a brass lamp, windows made of silica, many things made of plastic derived from petrochemicals, fireplace bricks derived from clay (kaolinite), a tin cup, a gold wedding ring, walls made of plasterboard comprised of gypsum, steel nails, and screws in the furniture and paint on the walls containing diatomite as filler.
Minerals Used in Everyday Life: At The Hardware Store
Take a field trip for a scavenger hunt at a hardware store. To get started, here are a few things to seek:
• aluminum and tin siding or roofing (from bauxite or cassiterite)
• bricks and ceramic products (from fired clay, or kaolinite)
• diatomaceous earth for swimming pool filters
• drill bits and saw blades used for cutting tile, concrete, etc. (from diamond)
• electrical wiring, pipes, and plumbing fixtures (from copper)
• glass (from silica sand)
• plaster and drywall (from gypsum)
• rough and crushed rocks and stones for ornamental use (scoria, limestone, marble, etc.)
• sand for mixing with concrete, for sandboxes, etc.
• slabs of various sorts (granite, marble, etc.) for kitchen countertops
• steel and iron nails (made from iron ores like hematite)
Everyday Science with Minerals: Magnetite in ActionTo illustrate a practical use of a mineral, consider magnetite, or the magnetic version of iron ore. Both natural and synthetic magnets have negative and positive poles that cause them to attract or repel one another. A positive pole on one magnet attracts a negative pole on another magnet, and this attraction brings the two together. But two negative or two positive poles will push magnets apart. One neat result? Levitation! Certain train systems use this phenomenon to help trains move at higher speeds. To see a very practical effect of the mineral world, try this with so-called “doughnut” magnets on a stick that magically float one above another. |
Start a Collection of Everyday Minerals
Entire collections can be made of the raw materials of our built environment. Many common minerals are inexpensive and readily available from show dealers. As a start, consider pennies and a copper nugget; nails and hematite; fluorinated toothpaste and a fluorite crystal; laundry detergent and borate minerals; table salt and halite crystals; matches and sulfur.

Resources to Learn About Minerals in Daily Life
Several websites provide handy tables linking minerals to everyday objects. Here’s a sampling:
• Minerals Education Coalition
• United States Geological Survey (USGS)
• American Geosciences Institute (AGI)
• Gemological Institute of America (GIA)
• AFMS Future Rockhounds of America Badge Manual
The Role of Minerals in Human History

As we humans progressed from the Stone Age to the Electronic Age, we’ve seen all sorts of ages in between dominated by a search for earth resources. Consider gold rushes, wars of conquest for mineral-rich colonies, and “titans of industry” (Carnegie, Rockefeller, Peabody, Getty). Our current age is obsessed with a quest for minerals for electric batteries built with lithium and cobalt. These resources are eagerly being sought to move us from a carbon-emitting, petroleum-dependent economy to one based on clean energy.
However, keep in mind that clean electric energy still requires dirty mining. If you think we can get to a so-called no-cost energy future, think again! There will always be a need for mining and minerals, along with a cost to pay. How we ultimately balance such costs is what matters. Think we can live without minerals and all that goes into extracting them? Think again. Think wisely.
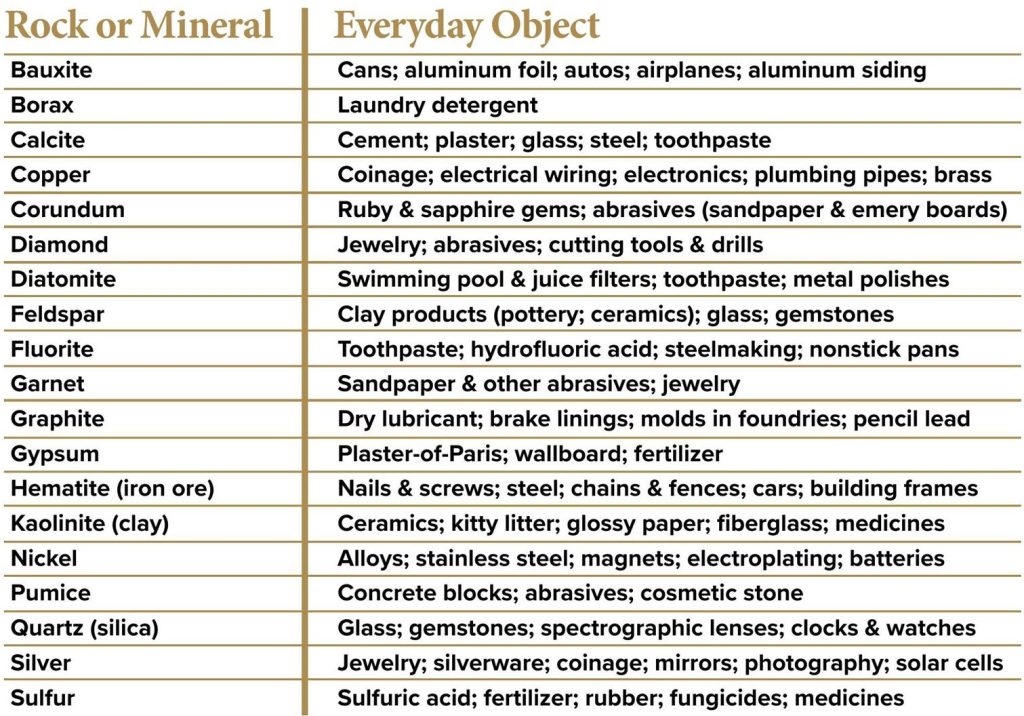
Everyday Objects Made from Minerals
Pencils or smartphones are just the beginning. There are thousands of minerals and even more applications of those minerals. Here’s a tiny selected sampling…
|
|
Minerals Used in Everyday Life: Final Thoughts
From pencils to smartphones, minerals used in everyday life touch nearly everything we see and use. They build our homes, power our technology, and even shape human history. Whether you’re spotting them in household items, exploring them in a scavenger hunt, or adding them to a collection, minerals reveal the hidden connections between the natural world and daily living. The next time you reach for something as simple as a light bulb or a salt shaker, take a moment to appreciate the minerals that make modern life possible.
This story about the minerals used in everyday life previously appeared in Rock & Gem magazine. Click here to subscribe. Story by Jim Brace-Thompson.